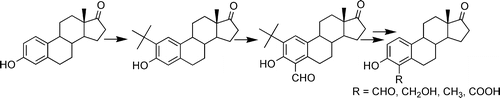
1. Synthesis of 4-Formyl Estrone Using a Positional Protecting Group and Its Conversion to Other C-4-Substituted Estrogens
4-Formyl estrone was synthesized in overall good yield in three steps starting from estrone. This was achieved by conducting an electrophilic aromatic substitution reaction using formaldehyde, triethylamine, and MgCl2 on 2-tert-butyl estrone, which was readily prepared in 96% yield from estrone using tert-butyl alcohol and BF3OEt2. The tert-butyl group acted as a positional protecting group to prevent reaction at the 2-position. The tert-butyl group was readily removed in good yield using AlCl3 in dichloromethane/CH3NO2. To our knowledge, this represents the first use of a positional protecting group for the synthesis of a C-4-modified estrogen. 4-Formyl estrone was used as a common precursor to obtain a variety of other C-4 modified estrogens in very high yields such as 4-methylestrone and 4-hydroxymethylestrone as well as the novel estrogen 4-carboxyestrone. The syntheses of 4-formyl, -methyl-, and -hydroxymethyl estrone represent dramatic improvements over previously reported syntheses of these compounds.
Download PDF »
View on PubMed »
Liu, Y.; Kim, B.; Taylor, S. D. J. Org. Chem. 2007, 72, 8824–8830.